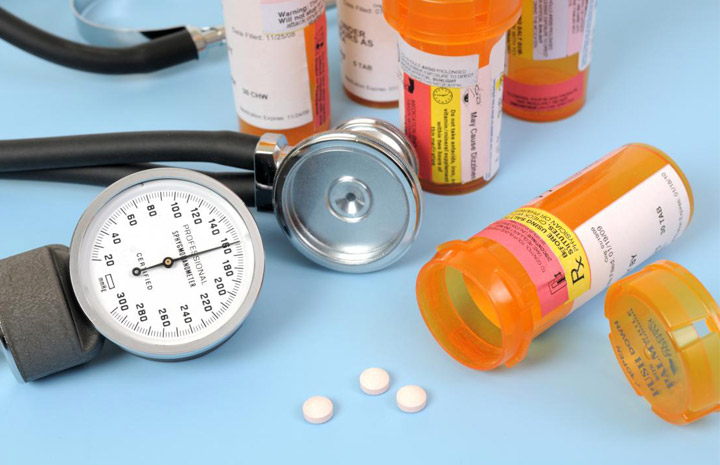
If you’re a blood pressure patient and regularly taking some pills to maintain your blood pressure, then you might want to stop- taking those pills if you don’t want to get cancer. Because according to the U.S. Food and Drug Administration, there are some medicines which they have recalled because of the impurity they contained which might cause cancer to its consumer. FDA said they found some contamination: N-nitrosodiethylamine (NDEA) in some of these drugs and that’s they have recalled them to prevent any further consumption of these contaminated drugs.
Irbesartan tablets manufactured by ScieGen Pharmaceuticals and labeled as Westminster Pharmaceuticals and Golden State Medical Supply, Inc (GSMS) have been recalled after it’s been found out that these medicines contain an impurity. FDA has also published a complete list of those effective drugs which they have recalled and most of these drugs used to sold in 75 mg, 150 mg, and 300 mg doses. Irbesartan drug is mostly used by those patients who have got high blood pressure and in some cases to cure kidney diseases too, but since enow FDA has announced this drug impure, it’s advisable not to consume any pills of it.
However, it’s a bit more surprising because this type of thing had happened earlier year also when the same contamination was found in some blood pressure medicines and at that time more than 55 medications were recalled. According to the FDA Nitrosodiethylamine is a compound which is mostly found in nature but it also has been traced into the manufacturing process, and it’s a part of air pollution, and that’s why containing any medicine in which this compound is included could be very dangerous for any patient. However, if you have any drugs in your consumption list which got affected by this recall, then it’s advisable for you to get alternative medicine under doctor’s prescription.

Jeffrey is acting editor in chief of AmazingNews24 with over seven years of experience in the field of online news under his belt. Jeffrey has worked with multiple media houses and is currently leading a team of journalists, sub-editors and writers through his entrepreneurial endeavours.